Rationale for the Use of Irradiated Blood Irradiation of cellular blood components is a well-established intervention for the prevention of transfusion-associated graft-versus-host disease TA-GVHD. Irradiation of blood components is a well-established intervention for the prevention of transfusion-associated graft-versus-host disease TA-GVHD.
Https Nhsbtdbe Blob Core Windows Net Umbraco Assets Corp 15547 Blc6863 Irradiated Blood Factsheet Hi Res Pdf
Who doesnt need irradiated products.
Irradiated blood products indications use. ATG we now recommend use of irradiated blood components for aplastic anaemia patients receiving immunosuppressive therapy with ATG andor alemtuzumab 2C. Irradiated blood components indicated after alemtuzumab anti-CD52 therapy. Cells post irradiation and practices involving irradiation of autologous blood collected by intraoperative cell salvage were consulted.
As new potent immunosupressive drugs and biological agents are introduced into practice there is a need for regular review of these recommendations 2C. Top Up Transfusions Neither premature nor term infants require irradiated blood products for top-ups even multiple top ups. Epub 2020 Aug 18.
Why is it important these patients receive irradiated blood components. Irradiated products will be provided until T Lymphocyte immunodeficiency syndrome has been excluded. Multiple sclerosis and vasculitis.
Multiple sclerosis MS vasculitis and. 10 rows Normally there is a warning sticker on the front of the patients casenotes and. Indications for irradiated blood products To prevent transfusion associated GVHD in vulnerable patients the following groups of patients require irradiated blood products.
Other indications Not Haemato-Oncology IUT Neonates who have previously had an IUT Recipients of blood products donated by a relative. In the absence of other indications as listed above autologous bone marrow or stem cell transplant patients require irradiated components for 3 months post transplant 6 months if total body irradiation is used for conditioning. We cannot make a rm recom-mendation as to how long irradiated components should continue to be used after ATG administration.
Their use after rituximab antiCD20 is not recommended at this time. Irradiated components Irradiated blood components are used to prevent Transfusion-associated graft-versus host disease TA-GVHD the primary cause of which is proliferation and engraftment of transfused donor T-lymphocytes in the bone marrow of susceptible recipients. Authors Theodora Foukaneli 1.
Why is Blood Irradiated. TA-GVHD a rare complication fewer than one per million transfusions result in. Irradiating blood components prevents the donor white cells replicating and mounting an immune response against a vulnerable patient causing.
Transplant patients from onset of conditioning for life including high dose therapy with autologous rescue. Irradiated blood components should be used after alemtuzumab antiCD52 therapy. It is prudent to offer irradiated components to these patients.
Irradiated blood components should be used for 3 months after receiving CART therapy unless the situation indicates that it should be continued indefinitely. As described in the Technical Manual 20th Edition and Circular of Information October 2017 cellular blood components are irradiated prior to transfusion to prevent the proliferation of viable T lymphocytes which are the immediate cause of Transfusion Associated-Graft Versus Host Disease TA-GVHD. Guidelines on the use of irradiated blood components.
Patients at risk of TA-GvHD should be made aware of. To provide healthcare professionals with clear guidance on situations when the use of irradiated blood components is indicated. There is a system cost of providing irradiated blood components to patients that includes higher production costs time delays and damage to.
G2C Recommendation The situation with other purine antagonists such as bendamustine and clofarabine is unclear but use of irradiated blood components is recommended as these agents have a similar mode of action. Use of irradiated cellular blood components for patients receiving immunosuppressive agents ATG or alemtuzumab for non-haematological indication. Guidelines on the use of irradiated blood components Br J Haematol.

Transfusion Of Blood And Blood Products Indications And Complications American Family Physician
Lymphoma Action Blood Transfusions

Blood Transfusion Irradiation Of Blood Products

Transfusion Of Blood And Blood Products Indications And Complications American Family Physician

Pdf Transfusion Of Blood And Blood Products Indications And Complications Semantic Scholar

Blood Products And Their Clinical Applications And Specifications Download Table

Why Do We Irradiate Blood Youtube

Patients Requiring Irradiated Blood Components Download Table
Http Jdc Jefferson Edu Cgi Viewcontent Cgi Article 1062 Context Patientsafetyposters

Irradiation Of Blood And Blood Components Ppt Download
Https Www Albertahealthservices Ca Assets Wf Lab Wf Lab Clin Tm Irradiated Use Pdf

Indications For Irradiated Blood Transfusion Download Table

Blood Products And Their Clinical Applications And Specifications Download Table

Table 4 From Transfusion Of Blood And Blood Products Indications And Complications Semantic Scholar
Blood Transfusion Support In Stem Cell Transplant Salwa

Blood Components Professional Education
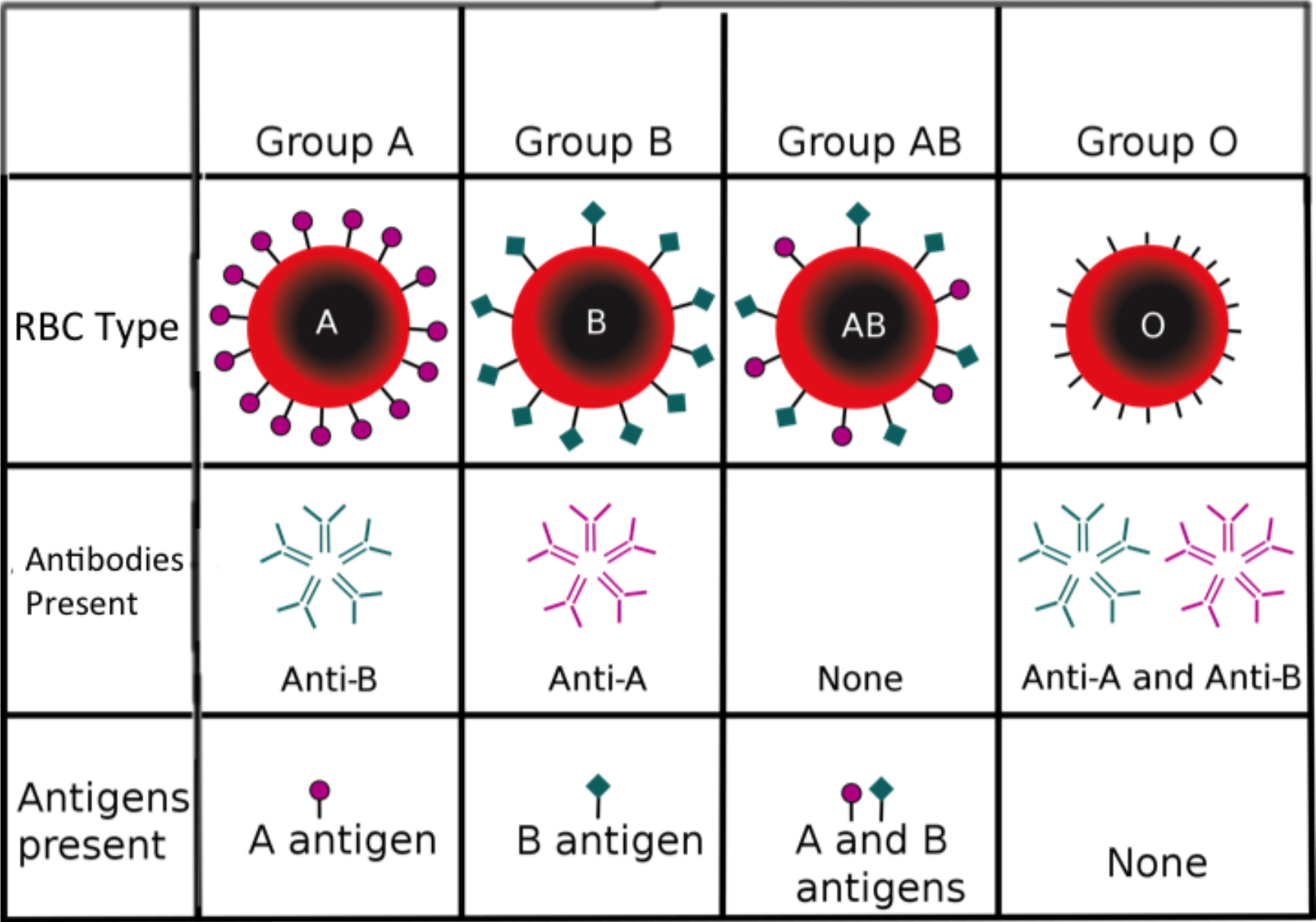
Bood Transfusion Part 1 Basics Anaesthesia Tutorial Of The Week 242 Wfsa Resources


