A state or a condition of an atom or a molecule having positive and also negative charges especially in case of magnetic or an electrical poles. A molecule is polar when there is a non-uniform positive and negative charge to the atom.
Factors Affecting Solubility Mrs Thompson
Lets take a look at a classic example of polarity.
What is polar mean in chemistry. Polar is a term used to describe a type of covalent bond that happens when electrons are shared between two different atoms. Let us understand the concept of polar and non-polar solvents. Several classes of molecules have simultaneously both hydrophilic and hydrophobic characteristics amphiphilic molecules with a polar head interacting favourably with water and a non polar tail which escapes the contact with water.
By definition polarity is basically the status of having poles In a molecule this means that the molecule has poles of positive and negative chargealso known as a separation of charge. Polar solvent is a type of solvent that has large partial charges or dipole moments. A polar molecule is formed when one end of a molecule has a positive charge and the opposite end has a.
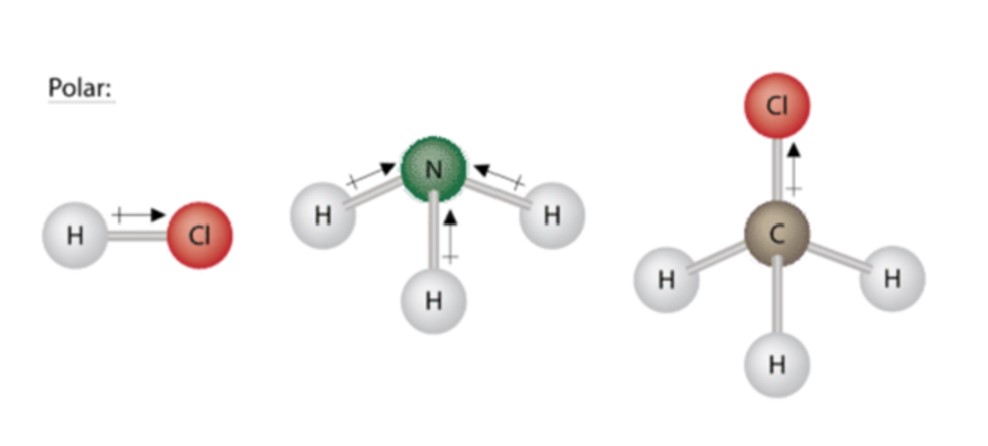
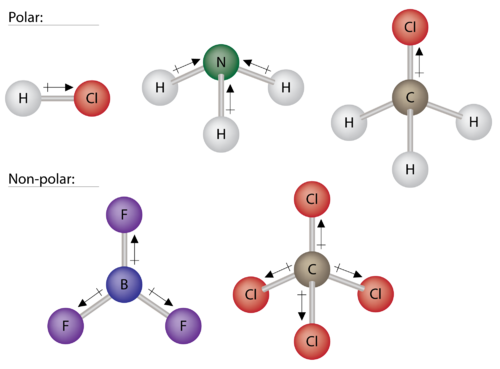
Due to the non-symmetrical shape of the molecule bent the molecule itself is polar it has a. This results in shared electrons spending more time on the side with greater electronegativity causing that. The more electronegative an atom the more it seeks electrons.
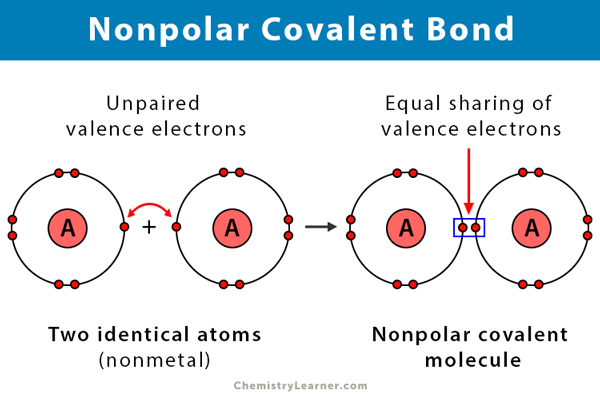
Both of the bonds inside the molecule are polar bonds. Monolayer of a fatty acid on water. While bonds between identical atoms such as two of hydrogen are electrically uniform in that both hydrogen atoms are electrically neutral bonds between atoms of.
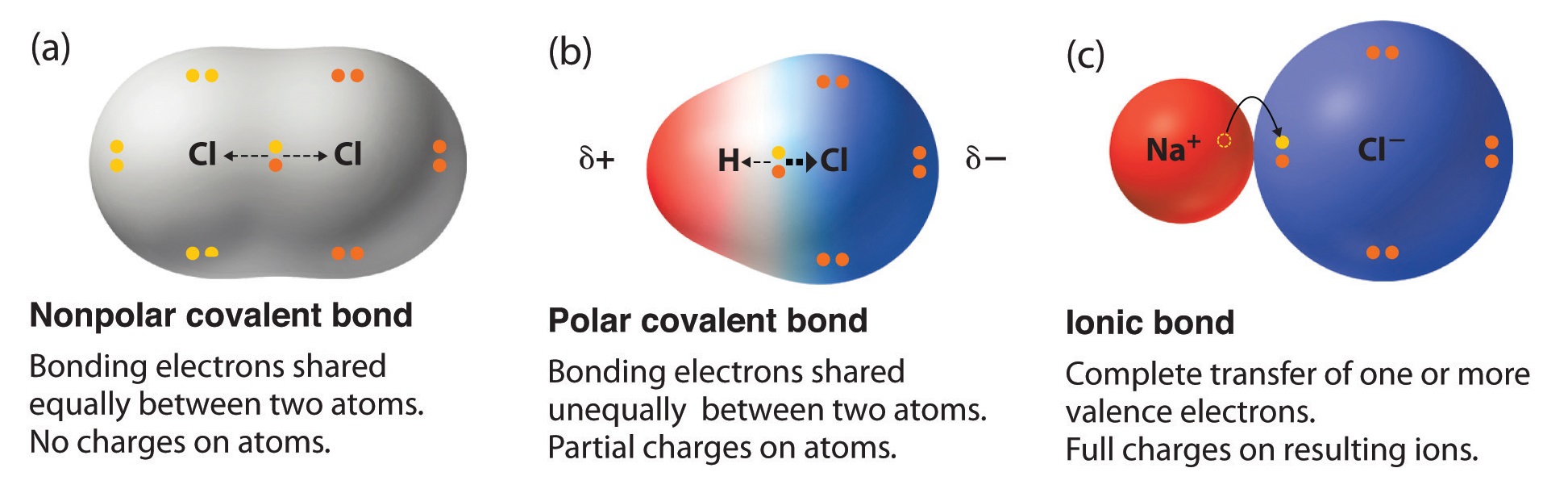
A molecule can be ionic polar or non-polar. In chemistry polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole moment with a negatively charged end and a positively charged end. Polarity in general refers to the physical properties of compounds such as boiling point melting points and their solubilities.
They form when the electronegativity difference between the anion and cation is between 04 and 17. This occurs in molecules that are asymmetric along at least one axis when one side contains atoms with a greater electronegativity than the other side. Water molecules are polar molecules.
Polar solvents can be further divided into protic and aprotic. Polar bonds are usually liquids or solids and are soluble. Polarity is a physical property of compounds which relate to other physical properties such as melting and boiling points or solubility.
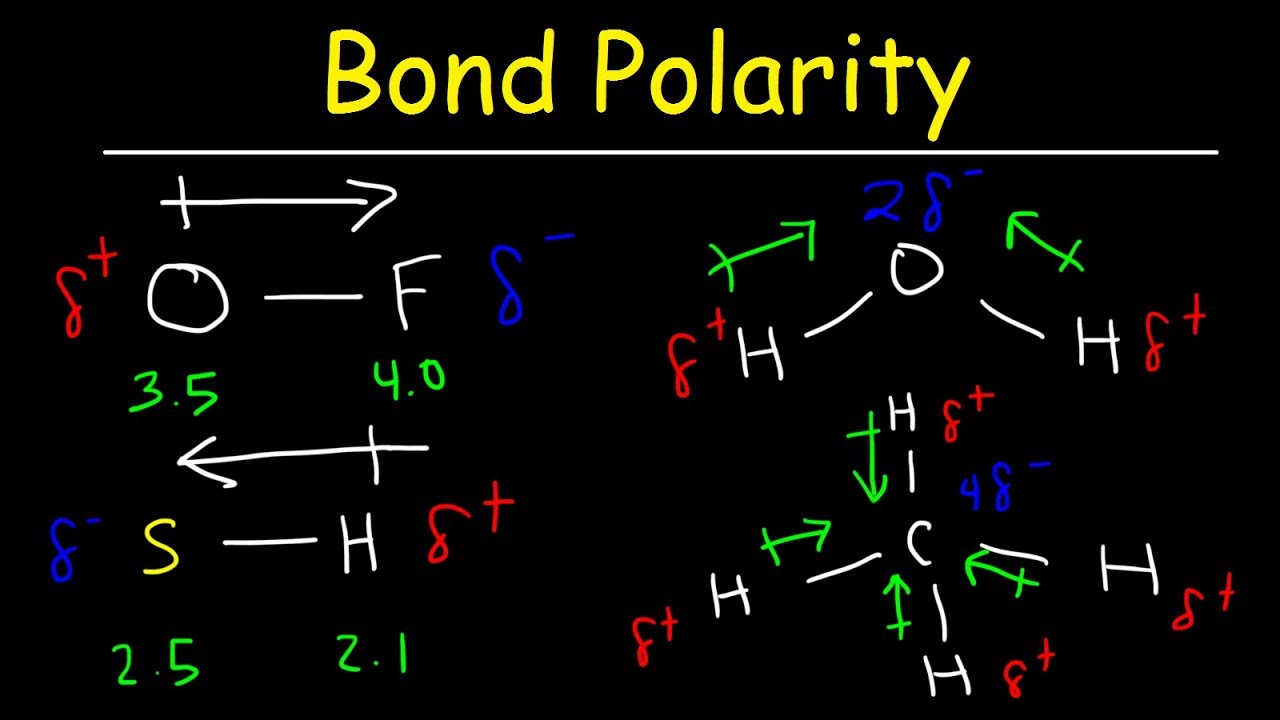
Polar bonds form when there is a difference between the electronegativity values of the atoms participating in a bond. This is caused by the geometry of the atoms in the molecule causing more electronslone pairs to positioned on one side than the other. A polar bond is a type of covalent bond in which the electrons forming the bond are unequally distributed.
Polar bonds are intermediate between pure covalent bonds and ionic bonds. A polar molecule is a particle consisting of two or more covalently bonded atoms with an asymmetric distribution of charges. Polar molecules must contain polar bonds due to a difference in electronegativity between the bonded atoms.
Bond polarities arise from bonds between atoms of different electronegativity. For example H2O is a. The bonds between the atoms have very different but measurable electronegativities.
The theory of electronegativity lies in entire inorganic chemistry. Polarity and Non-polarity of molecules depend upon the electronegativity. Polarity in chemical bonding the distribution of electrical charge over the atoms joined by the bond.
If one atom is more electronegative than others it can form an ionic bond or a polar covalent bond. Its important to note here that in order for a solution to form the molecules must be either both polar or both non-polar. A polar molecule is a molecule containing polar bonds where the sum of all the bonds dipole moments is not zero.
A non-polar molecule is fat soluble. Polar protic solvents are water ethanol methanol ammonia acetic acid and others. Polar in chemistry also know as a polar covalent bond happens when 2 or more non-metals create a bond.
The formation of an ionic bond happens when a major electronegative atom takes an electron. In other words the electrons spend more time on one side of the bond than the other. Polar protic solvents are capable of hydrogen bonding because they contain at least one hydrogen atom connected directly to an electronegative atom such as O-H or N-H bonds.
Use water as an example of a mol. A polar molecule is water soluble.
Chemistry Polar And Non Polar Molecules

Polar And Nonpolar Molecules Youtube

What Is Polarity Definition Example Polar Vs Non Polar Molecules
4 12 Shapes And Properties Polar And Nonpolar Molecules Chemistry Libretexts

Polar Molecules Chemistry For Non Majors

Bonds Shmoop Biology Chemistry Classroom Science Chemistry Chemistry Education

Water Molecules Are Polar In Nature Water Molecules Are Basically H 2o Molecules Which Have Bent Shapes Also O Atom Water Molecule Molecules Hydrogen Bond
Illustrated Glossary Of Organic Chemistry Polar Nonpolar
Polar And Nonpolar Molecules Youtube

Chemical Bonding Definition Examples And Importance In Chemistry Chemsolve Net Chemistry Electron Configuration Chemical Bond

Bromine Electron Configuration Electron Configuration Element Symbols Element Chemistry
Polar Molecules Chemistry For Non Majors

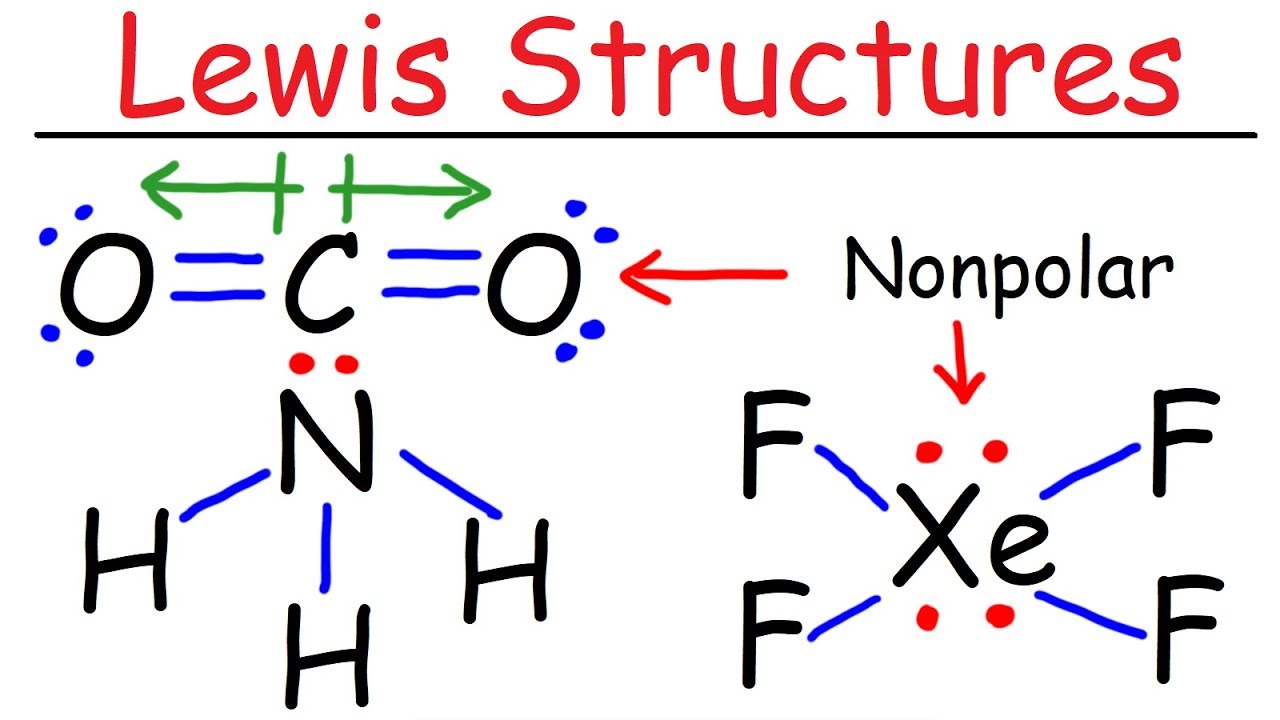
Simple Method For Writing Lewis Structures Ozone O3 And Carbonate Co3 2 Molecular Geometry Writing Systems Biology

What Is Polarity Definition Example Polar Vs Non Polar Molecules

Molecule Vs Atom Cengage Learning Molecules Teaching Science

Inert Pair Effect Definition Examples Cause And Consequences Chemsolve Net Electron Configuration Physical And Chemical Properties Chemical Bond
Bond Polarity Electronegativity And Dipole Moment Chemistry Practice Problems Youtube
Nonpolar Covalent Bond Definition And Examples

9 3 Molecular Shape And Molecular Polarity Chemistry Libretexts
